Introduction: Lack of factor VIII/IX (FVIII/FIX) in hemophilia A/B (HA/HB), respectively, results in reduced thrombin generation, leading to recurrent/spontaneous bleeds. Concizumab is an anti-tissue factor pathway inhibitor (TFPI) monoclonal antibody, currently under clinical investigation for subcutaneous prophylaxis of HA/HB patients with/without inhibitors. Breakthrough bleeds occurring in HA/HB patients while on concizumab prophylaxis may be treated with FVIII/FIX. We aimed to compare the in vitro effect of recombinant FVIII (rFVIII) and FIX (rFIX) in HA and HB plasma, respectively, in the absence or presence of concizumab.
Methods: rFVIII/rFIX was added to HA/HB pooled plasma at 0.25, 0.5 or 1 IU/mL (corresponding to post-administration plasma concentrations of 12.5, 25 and 50 IU/kg rFVIII and 12.5−25, 25−50 and 50−100 IU/kg rFIX) in the absence or presence of concizumab (1,500, 4,500 or 15,000 ng/mL). In a separate experiment, 33 plasma samples from eight HA patients, who were on concizumab prophylaxis as part of the phase 2 explorer5 trial (NCT03196297), were spiked with 0.5, 1 and 1.5 IU/mL rFVIII. Pre-dose samples (before concizumab prophylaxis) from seven of these patients were also included. Thrombin generation was measured after initiation with 1 pM tissue factor (PPP-Low, Thrombinoscope). Statistical analysis of the effects conferred by each (combination of) drug(s) was performed by ANOVA analyses.
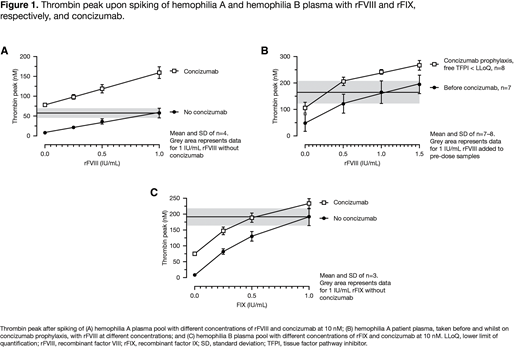
Results: A significant (p<0.001) and concentration-dependent increase in thrombin peak was observed when HA plasma pool samples were spiked with rFVIII, both in the absence and presence of concizumab. Likewise, concizumab increased the thrombin peak both in the absence and in presence of rFVIII. Increasing concizumab from 1,500 to 4,500 and 15,000 ng/mL only slightly increased the thrombin peak further, demonstrating that a close-to-maximal effect on thrombin peak was achieved at 1,500 ng/mL concizumab. The effects of concizumab and rFVIII were mainly additive with an up to 20% additional effect caused by drug-drug interaction. The addition of rFVIII to explorer5 patient plasma samples resulted in a significant and concentration-dependent increase in thrombin peak. The effects observed for rFVIII and concizumab were exclusively additive. The thrombin peak obtained with 1.0 IU/mL rFVIII before concizumab administration was lower than with 0.5 IU/mL rFVIII in the presence of concizumab. This suggests that a 2-fold reduced rFVIII dose may be sufficient to achieve the same plasma thrombin generation capacity as with the standard rFVIII dose in the absence of concizumab. The addition of rFIX to a HB plasma pool increased the thrombin peak significantly (p<0.001) and in a concentration-dependent manner both in the absence and presence of concizumab (1,500 ng/mL). Likewise, concizumab increased the thrombin peak at all rFIX concentrations (p<0.001). Increasing concizumab from 1,500 to 4,500 and 15,000 ng/mL had no or limited further effect. The effects of concizumab and rFIX were mainly additive with an up to 10% effect conferred by negative drug-drug interaction for 1 IU/mL rFIX combined with concizumab >1,500 ng/mL and 0.5 IU/mL rFIX combined with 15,000 ng/mL concizumab, i.e., a 10% smaller effect of rFIX was observed in the presence of concizumab than in its absence. The thrombin peak obtained upon adding 1.0 IU/mL rFIX to plasma without concizumab was similar to the thrombin peak in the presence of concizumab and 0.5 IU/mL rFIX. This suggests that in the presence of concizumab, a 2-fold reduced dose of rFIX would be sufficient to obtain the same plasma thrombin generation capacity as with 1.0 IU/mL rFIX in the absence of concizumab.
Conclusion: rFVIII/rFIX increased the thrombin peak in HA and HB plasma, respectively, both in the absence and presence of concizumab. The combined effects of rFVIII/rFIX with concizumab were mainly additive with an up to 20% additional effect caused by drug-drug interaction with rFVIII and a 10% reduction with rFIX. No signs of exaggerated thrombin generation were observed by combining concizumab with rFVIII/rFIX. Therefore, the data support rFVIII/rFIX use for bleed treatment in patients on concizumab prophylaxis. As rFVIII/rFIX and concizumab have additive effects in terms of thrombin generation capacity, data suggest that clinical effectiveness could be achieved with rFVIII/rFIX doses in the lower range recommended for such products.
Kjalke:Novo Nordisk A/S: Current Employment, Current equity holder in publicly-traded company. Andersen:Novo Nordisk A/S: Current Employment, Current equity holder in publicly-traded company.
Author notes
Asterisk with author names denotes non-ASH members.

This feature is available to Subscribers Only
Sign In or Create an Account Close Modal